公司新闻
Company News
新闻中心 | 公司新闻 9366次阅读 2022-05-03 10:29
近日,郑州大学第一附属医院张明智教授团队和博生吉医药科技(苏州)有限公司(以下简称“博生吉”)研发团队在Clinical Cancer Research杂志上联合发表了非基因编辑手段制备的CD7-CAR-T(PA3-17注射液)治疗复发难治T-ALL/T-LBL的一项突破性成果。与以往发表的同种异体CD7-CAR-T或是供者来源的CD7-CAR-T治疗策略不同的是, PA3-17注射液是一款靶向CD7的自体CAR-T单药疗法。

在这项研究中,展示了8例患者的长期随访情况。结果显示,87.5%的患者既往治疗线数>4线,入组后均接受了PA3-17注射液的单次输注治疗(仅1例进行了两次回输),输注剂量为以下三种剂量水平之一: 1×106 CAR-T细胞/kg、1.5×106CAR-T细胞/kg或2×106 CAR-T细胞/kg。在8例接受PA3-17注射液输注的患者中,6例患者(75.0%)在第30天达到缓解,3个月时缓解率增加至87.5%(7/8); 其中,1例患者保持CR状态超12个月;其余患者均保持至少3个月以上的CR状态;
此外,对于T淋巴母细胞淋巴瘤累及骨髓的患者(T-ALL/LBL),患者在接受PA3-17注射液治疗后,淋巴瘤经PET-CT或CT扫描确认后,所有淋巴瘤病灶(涉及颈部,腋窝,腹膜等多处部位)均达到CR状态;骨髓经骨髓涂片及多色流式检测,也均达到CR且MRD-的状态。
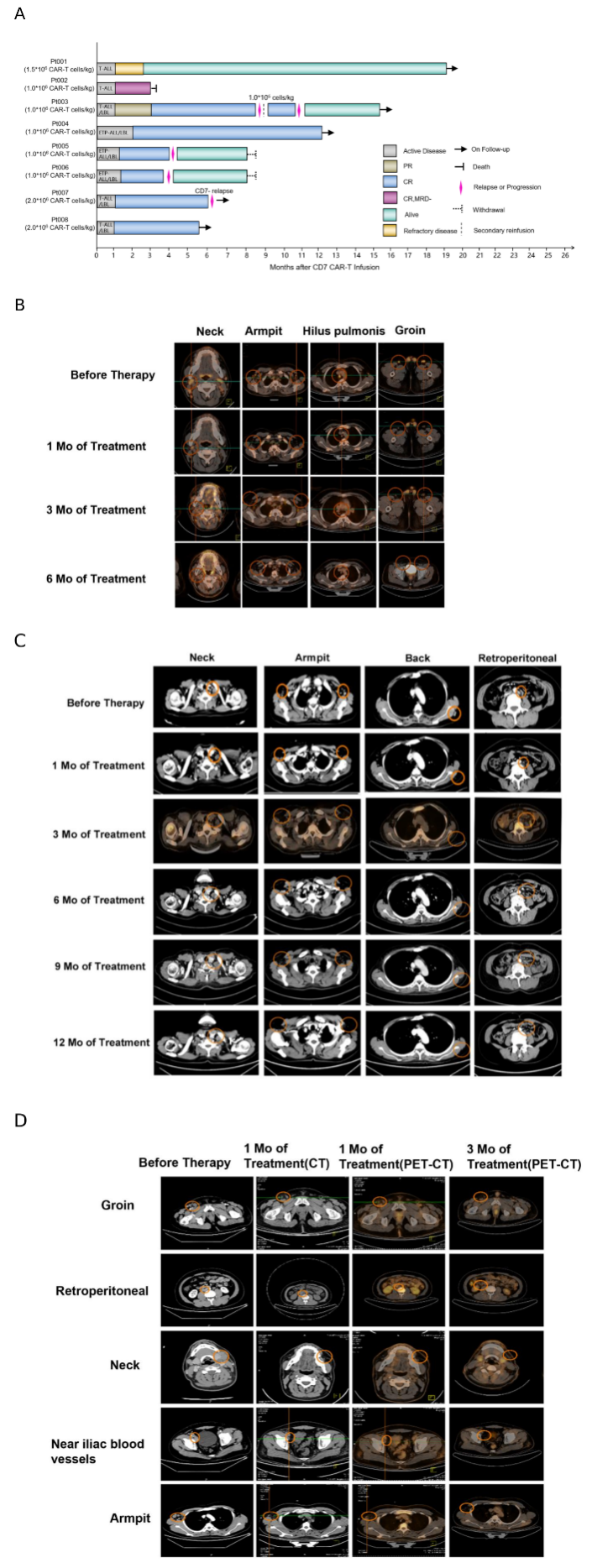
▲r/r T-ALL/LBL患者输注PA3-17注射液后的影像学疗效评估(图片来源:文献1)
CAR-T细胞体内扩增以及持久性是CAR-T疗效的关键性指标,从患者的长期随访数据来看,
CAR-T细胞在8例患者体内均产生了有效扩增,在回输12天左右, CAR-T细胞在体内扩增达到峰值,中位拷贝峰值为857.2 cells/µL,甚至在回输后的270天依然能够在患者体内检测到CAR-T细胞的存在,证实了PA3-17注射液在体内的良好持久性。
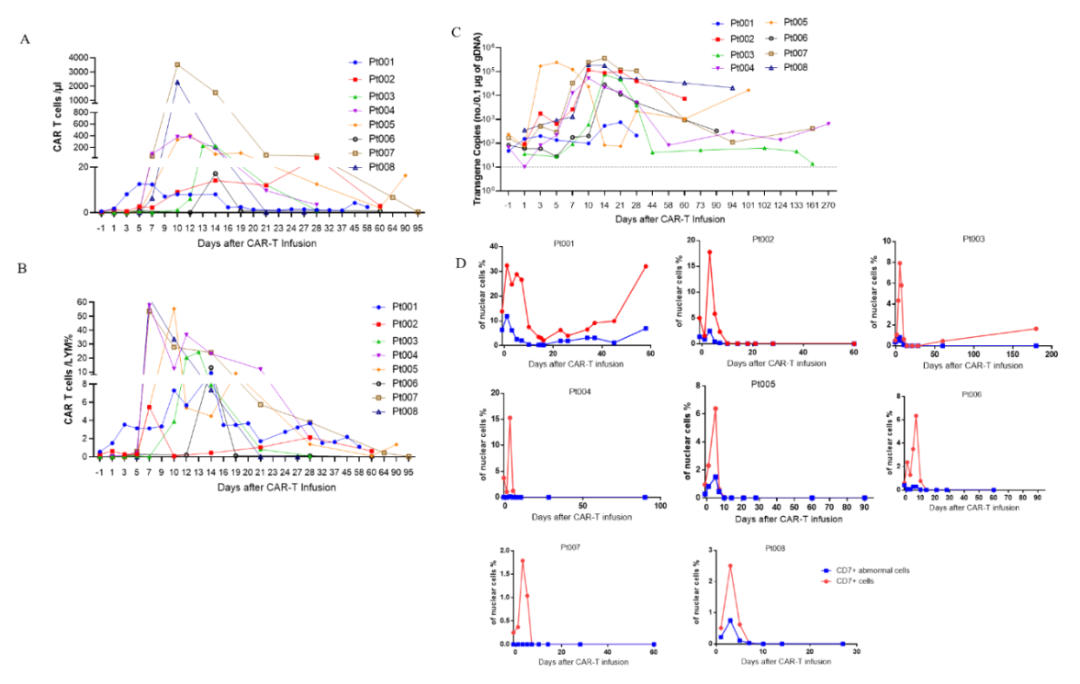
▲r/r T-ALL/LBL患者输注PA3-17注射液后的PK和PD分析(图片来源文献1)
所有8例患者均对PA3-17注射液的单次输注耐受良好,且未观察到神经毒性事件(ICANS)。尽管8例患者都出现了不同程度的细胞因子释放综合征(CRS),但大部分患者(7例,87.5%)的CRS为1级或2级。另外,在本研究中,从患者的长期随访数据可以看到,回输CAR-T细胞后虽然会出现短暂的患者正常T细胞被清除的现象,但患者T细胞很快会恢复,一方面是回输的自体CD7-CAR-T细胞在患者体内的增殖,另一方面患者体内CD7阴性的T细胞会代偿性增殖,最终所有患者都未产生明显的T细胞缺乏。

▲患者T细胞在输注PA3-17注射液后均有所恢复(图片来源:文献1)
小结:
r/r T-ALL/T-LBL是一种侵袭性极强的恶性血液肿瘤,复发难治的患者预后更差,治疗方式选择非常有限,5年以上生存率小于10%。此次PA3-17注射液相关研究的长期随访数据结果喜人,预示着该候选产品有望成为一种极具潜力的CAR-T细胞单药疗法。相信在未来,能够进一步拓展T-ALL/LBL以外的其他适应症,满足更多临床未满足需求,造福更多患者。
关于T-ALL/LBL
T淋巴母细胞白血病(T-ALL,T cell acute lymphoblastic leukemia)是一种高度侵袭性的血液肿瘤。以大量未成熟淋巴母细胞侵犯骨髓为特征,T-ALL的标准治疗包括放/化疗和干细胞移植。标准化疗方案只有30%-40%的响应率,且响应患者的中位总生存期为6个月。其中ETP-ALL存在更多的髓外浸润,并且保留了更多干细胞的特性,导致其治疗预后情况更差;
由于共有的表面抗原和潜在的恶性细胞污染,针对T-ALL的CAR-T细胞疗法发展滞后。另外,自2005年FDA批准奈拉滨(由GlaxoSmithKline完成商业化上市)以来,尚未有任何其他针对T-ALL的新疗法获批,而且2019年EHA(European Hematology Association)公布的关于奈拉滨治疗T-ALL/T-LBL的临床四期的数据显示患者的五年生存率只有18%,中位生存期为8个月。
关于PA3-17注射液
博生吉研制成功的PA3-17注射液,采用非基因编辑策略,在前期研究中表现出优异的药代和药效学数据,疗效显著,安全性较高。而且,由于开发成功高度优化的全自动制备工艺,成本显著降低,将有望成为普通百姓患者用得起的创新药物。目前,博生吉正在中国开展一项多中心的、由研究者发起的I期临床试验,旨在评估PA3-17注射液治疗复发难治性T-ALL /LBL患者的安全性及有效性,这也是国际首个基于CD7靶点的自体CAR-T注册临床试验。2022年4月19日,PA3-17注射液I期临床试验首例患者在郑州大学第一附属医院顺利完成回输。
关于博生吉
成为创新细胞药物研发的领跑者是博生吉的愿景,围绕临床未满足需求、开发患者真正需要的创新药物是博生吉的使命。博生吉是一家以突破性肿瘤细胞免疫治疗技术与细胞药物产品研发为主要发展目标的国家级高新技术企业。公司专注于开发具有国际领先水平的突破性First-in-class和Best-in-class CAR-T细胞药物,以造福肿瘤病患。
文献:
1. https://aacrjournals.org/clincancerres/article-abstract/doi/10.1158/1078-0432.CCR-21-4097/694479/Autologous-nanobody-derived-fratricide-resistant?redirectedFrom=fulltext